Credit: Chem (2023). DOI: 10.1016/j.chempr.2023.10.016
Phosphorus chemist Prof. Jan J. Weigand from the Dresden University of Technology, in collaboration with an interdisciplinary team, has developed a method to introduce phosphorus and nitrogen atoms into polycyclic molecules. This method holds the potential to pave the way for the development of new materials with specific optoelectronic properties, ideal for applications in organic semiconductor technologies such as OLEDs and sensors. The results were published this week in Chem.
Polyaromatic hydrocarbons, abbreviated as PAHs, play a central role in numerous (opto-) electronic applications, including chemical sensors, organic light-emitting diodes (OLEDs), organic field-effect transistors (OFETs), and organic solar cells.
Researchers are continually exploring the substitution of various elements beyond traditional carbon to optimize device performance and versatility. While substitution with boron (B), nitrogen (N), oxygen (O), and sulfur (S) has already undergone extensive research, the integration of phosphorus (P) in combination with nitrogen (N) remains a significant challenge.
Prof. Weigand and his research group at TUD Dresden University of Technology have recently achieved a significant breakthrough. "In our current research, we have developed an innovative method to selectively introduce phosphorus and nitrogen atoms into polyaromatic systems.
"This method allowed the synthesis of a wide range of P/N-substituted compounds, whose physicochemical properties were thoroughly investigated in collaboration with physicists from TUD. Through the combination of material simulations and spectroscopic measurements, we were able to gain fundamental insights into the structure-property relationships of the obtained compounds."
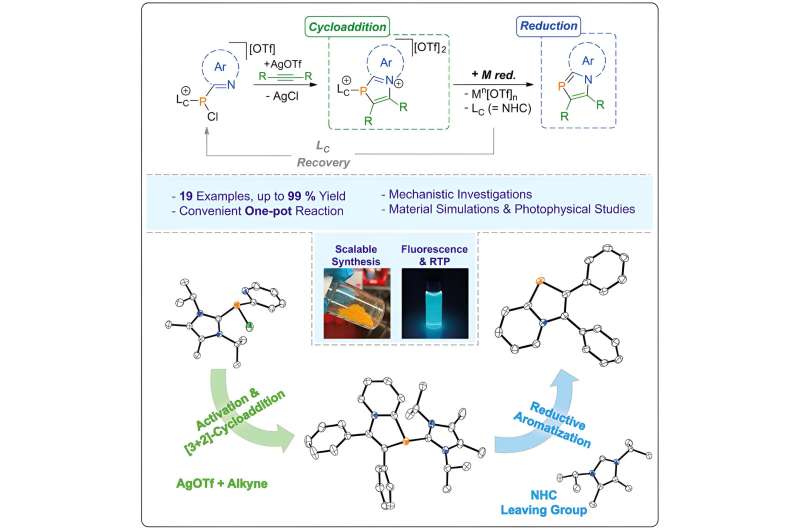
The new method provides access to the well-known class of azaphospholes, which were previously only accessible in a very cumbersome manner and mostly in very low yields. Therefore, they were not considered for (opto-)electronic applications until now.
"By deliberately combining phosphorus and nitrogen, we hope to be able to control the electronic and optical properties of these compounds in a way that was not possible before. This opens up exciting prospects for future applications in optoelectronics and beyond," adds Sebastian Reineke, head of the Light-Emitting and eXcitonic Organic Semiconductors Group (LEXOS) at TUD.
More information: Jannis Fidelius et al, Convenient access to π-conjugated 1,3-azaphospholes from alkynes via [3 + 2]-cycloaddition and reductive aromatization, Chem (2023). DOI: 10.1016/j.chempr.2023.10.016
Journal information: Chem
Provided by Dresden University of Technology